Join Our Groups
TOPIC 1: NON METALS AND THEIR COMPOUNDS



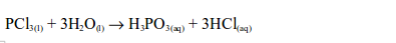

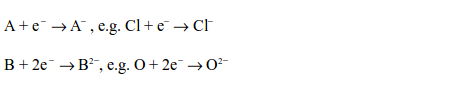


General Chemical Properties of Non Metals
The non-metals are very reactive and most of them react with other elements to form different compounds.
The following are important chemical properties of non-metals which are connected with their tendency towards electron gain in the course of formation of compounds:
1. The oxide of a non-metal is either acidic or neutral but never basic. The oxide of a non-metal is a covalent compound. Being acidic, it combines with water to form an acid, e.g.

2. A non-metal never replaces hydrogen in an acid to form a salt. This is because replacement of hydrogen in an acid is due to the fact that H+ accepts electrons supplied by a metallic atom.

A non-metal is an electron acceptor and so cannot release electrons to hydrogen ions in solution.
3. Non-metals form covalent chlorides, for example, the behaviour of phosphorus forming its chlorides is well known.

A covalent chloride like this is usually a volatile liquid, a non electrolyte, and rapidly hydrolysed by water.
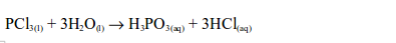
These properties are characteristic of non metallic chlorides (except CCl4 which is not hydrolysed by water).
4. Non-metals combine with hydrogen to form many hydrides. A covalent compound is formed by equal sharing of electrons between or among the combining atoms. For example, methane ammonia, hydrogen chloride and hydrogen sulphide are the covalent compounds.

5. Non-metals are oxidizing agents As discussed early, non-metals accept electrons from other substances. Therefore, they are called oxidizing agents because, upon accepting the electrons, the substances donating these electrons are oxidized. So they act as the agents for oxidation of other substances.
The Oxidizing Properties of Non-metals
Explain the oxidizing properties of non-metals
Non-metals react by gaining electrons to become negative ions. A univalent non-metal accepts one electron while a divalent one accepts two electrons. The ion formed carries the corresponding number of negative charges, but they rarely exceed two and never exceed three.
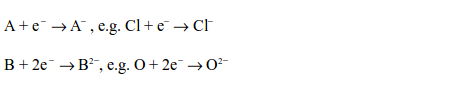
When a substance loses electron(s), it becomes oxidized, i.e. its oxidation number increases. This is called oxidation. Due to the fact that non-metals accept the electrons(s) donated by other substances, particularly metals, they are, therefore, termed as oxidizing agents. This is because by accepting the electrons, they help oxidize the electron donors.
Those substances or metals which donated the electrons are called reducing agents. This is because the electrons they donate reduce the oxidation number of non-metals. This process is called reduction. In this respect, non-metals act as oxidizing agents while metals act as reducing agents.
Strong and weak oxidants
As we have already seen, non-metals ionize by electron gain. In all cases, the extra electron(s) accepted lead to the formation of negative ions. The easiness of formation of negative ions depends on the ability of an element to accept the electrons. The ability of accepting electrons is called electronegativity of an element. Some elements are more electronegative than others.
The order of electronegativity of some non-metals is as follows: Fluorine > Chlorine > Bromine > Iodine > Nitrogen > Carbon. In this list flourine is the most electronegative and carbon is the least electronegative.
The degree of electronegativity indicates reactivity and hence oxidizing power of the element. Elements with higher electronegativity will displace those elements with lower electronegativity from their compounds.
Referring to the series above, fluorine will displace all the rest of the elements from their compounds as it is more electronegative than any other element in the series. Likewise, chlorine can displace bromine, iodine and nitrogen from their compounds. The displacement reaction occurs in the manner:

Where X is more electronegative than Y

The higher the electronegativity the stronger the oxidant. For example, bromine is a stronger oxidant than iodine, nitrogen and carbon.
The Displacement of Non-metals by another Non-metal from a Compound
Describe the displacement of non-metals by another non-metal from a compound
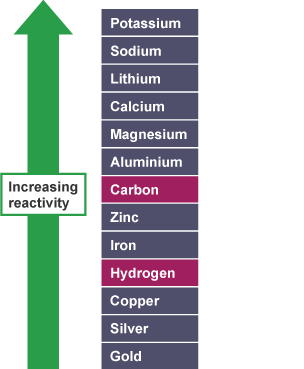
Non-metals in the reactivity series
It is useful to place carbon and hydrogen into the reactivity series because these elements can be used to extract metals.
Here is the reactivity series including carbon and hydrogen:

Note that zinc and iron can be displaced from their oxides using carbon but not using hydrogen. However, copper can be extracted using carbon or hydrogen.
Chlorine
Chlorine is very reactive, so it is never found as the free element in nature. It occurs mainly as sodium chloride or rock salt. It also occurs in the combined state as chlorides of sodium, potassium and magnesium.
How is chlorine made?
In industry, chlorine is made by electrolysis of molten sodium chloride or brine. Brine is a concentrated solution of sodium chloride in water.
In the laboratory, chlorine is made by the oxidation of concentrated hydrochloric acid. The oxidation can be brought about by a number of oxidizing agents, for example, lead (IV) oxide, manganese (IV) oxide, trilead tetraoxide (Pb3O4) or potassium manganate (VII).
The reaction equation is:
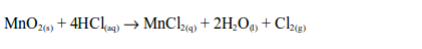
When potassium permanganate is used, reaction equation is:
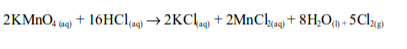
The poisonous nature of chlorine
Chlorine is a useful but dangerous gas. It is very poisonous if inhaled to even a small extent (1 part of chlorine in 50,000 parts of air causes death). Chlorine poisoning occurs when the gas is inhaled or swallowed. It reacts with water inside and outside of the body (such as water in the digestive tract and moisture on the lungs and eyes) to form hydrochloric acid and hydrochlorous acid. Both of these substances are extremely poisonous.
Most incidents of chlorine poisoning are due to ingesting household cleaners or bleach products. Due to its poisonous nature, chlorine was used in the World War I (1914-1918) to kill people and it caused many deaths.
The Chemical Properties of Chlorine
Describe the chemical properties of chlorine
1. When a little litmus solution is poured onto a gas jar of chlorine, litmus immediately turns colourless. The gas also bleaches a damp litmus paper since the gas is acidic. If blue litmus paper is used, it is first turned red before being bleached. The bleaching action is due to the fact that chlorine reacts with the water, forming a mixture of hypochlorous and hydrochloric acids.
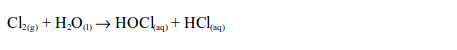
The hypochlorous acid is a very reactive compound and readily gives up its oxygen to the dye, to form a colourless compound:
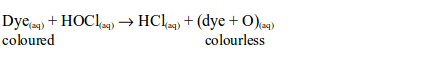
Thus, dry chlorine does not bleach. It has to be moist since the hypochlorous acid formed by its reaction with water is the one that is responsible for the bleaching action.
2. Chlorine reacts with many reactive metals, on heating, giving their respective chlorine salts, e.g.:
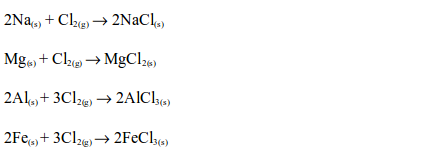
3. Some non-metals burn in chlorine to form covalent compounds:
(i) Dry, yellow phosphorous burns spontaneously in the gas to produce white fumes of chlorides of phosphorous, mainly PCl3.

4. When a filter paper dipped into a little turpentine is dropped into a gas jar of chlorine, a violent reaction occurs and a black cloud of solid particles of carbon is formed.
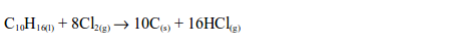
The reaction also takes place with other hydrocarbons as well and as with turpentine, hydrogen chloride gas is formed. The hydrogen chloride can be shown to be present by passing a little ammonia gas across the top of the jar whereby dense white fumes of ammonium chloride are observed.

5. Hydrogen chloride gas is oxidized to elemental sulphur by chlorine gas. When a gas jar of hydrogen sulphide is inverted over a gas jar of chlorine, a yellow precipitate of sulphur and hydrogen chloride gas will be formed.

6. When a stream of chlorine gas is bubbled through a pale green iron (II) chloride solution, a red-brown precipitate of iron (III) chloride is formed, showing that the iron (II) chloride has been oxidized to iron (III) chloride.
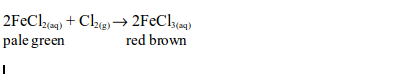
Ionically:
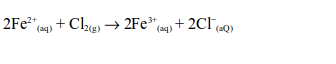
7. When chlorine is bubbled into the solution of sulphur dioxide in water for a few minutes, dilute sulphuric acid is obtained.
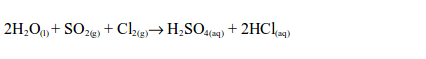
8. The reaction between chlorine and alkalis gives products which depend upon reaction conditions:
Chlorine reacts with cold dilute aqueous solution of sodium or potassium hydroxides, forming a pale yellow solution of the hypochlorite and chloride of the metal.

With the hot concentrated aqueous solution, a mixture of the chloride and chlorate is formed.
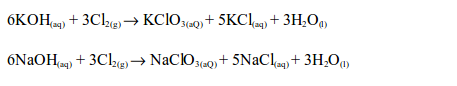
A similar reaction occurs if hot concentrated calcium hydroxide solution is used.
The Uses of Chlorine
Explain the uses of chlorine
1. Chlorine is bleaching agent and is also used in the manufacture of other bleaches. When chlorine is added to sodium hydroxide solution, bleach is made.
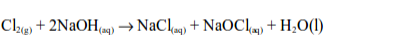
The active chemical in bleach is sodium hypochlorite (sodium chlorate (I))
If chlorine is passed for a considerable time over solid calcium hydroxide the product formed is bleaching powder.
Bleaching power finds extensive use in dye works, and in laundries. It is used in industries where cloth, cotton, paper, etc. need to be bleached. Many textile industries use chlorine for bleaching purposes. Bleach is also used to kill bacteria for example in the toilet. It will also remove colour from the dyed materials.
2. Chlorine is added to water supplied to homes and industries to kill disease-causing germs like bacteria. If they were not killed, these pathogens might cause diseases such as cholera and typhoid. It is also used to sterilize the water in swimming pools.
3. Chlorine is used to make some important chemicals such as hydrochloric acid, chlorofluorocarbons (CFCs), tetrachloromethane (CCl4), and chloroform (CHCl3).
CFCs are carbon compounds containing both chlorine and fluorine. An example is trichchlorofluoromethane (CCl3F).
CFCs are very uncreative compounds. They were used in aerosol cans and refrigerators. However, CFCs damage the ozone layer. Consequently, their use is strongly discouraged.
4. Chlorine is a reactant in the manufacture organo-chloro compounds which are used to make pesticides, antiseptics (e.g. dettol), herbicides (weed killers) and polyvinyl chloride (PVC) which is an intermediate compound in the production of plastics.
Hydrogen Chloride
A Dry Sample of Hydrogen Chloride Gas
Prepare a dry sample of hydrogen chloride gas
Hydrogen chloride is a gaseous compound at room temperature. It is usually prepared in the laboratory by the reaction between concentrated sulphuric acid and any chloride, e.g. sodium chloride. When a mixture of the two is gently warmed, hydrogen chloride gas is formed.
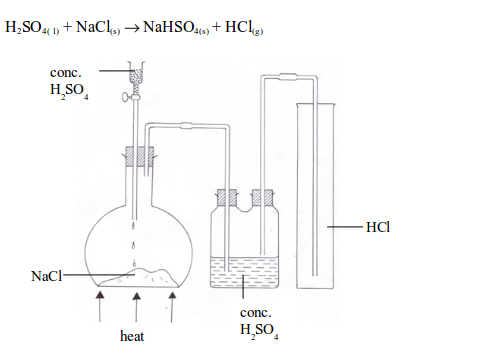
Preparation of hydrogen chloride gas
The Properties of Hydrogen Chloride Gas
Explain the properties of hydrogen chloride gas
(a) Physical properties
Includes
- It is a colourless gas with a choking, irritating smell and an acid taste.
- It is heavier (denser) than air.
- It fumes in most air due to the formation of tiny droplets of hydrochloric acid.
- It is very soluble in water (450 cm3 of gas in 1 cm3 of water). The acidic properties and the solubility of hydrogen chloride gas can best be shown by the fountain experiment
b) Chemical properties
1.A dry sample of hydrogen chloride gas has no effect on dry, blue litmus paper but it turns moist, blue litmus paper to red. This is due to acidic properties of hydrogen chlorine gas.
2. It does not burn and does not support combustion.
3. When a stream of hydrogen chloride gas is passed over some heated metals, the anhydrous chloride of these metals are formed:

4. It is decomposed by strong oxidizing agents such as manganese dioxide, lead dioxide and red lead to give chlorine gas.
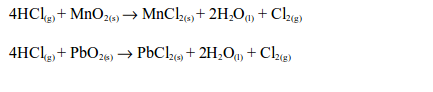
5. When the gas is passed though a solution of silver nitrate, acidified with dilute nitric acid, a white precipitate of silver chloride is formed. This is another test for hydrogen chloride gas and all soluble chlorides.

6. It gives clouds of white fumes when brought into contact with ammonia vapour. The fumes are solid ammonium chloride (NH4Cl).

In qualitative analysis, this is the chemical test for hydrogen chloride gas as well as for ammonia gas.
Reactions of aqueous hydrogen chloride
Hydrogen chloride gas is very soluble in water (and in other polar solvents). In water, an acid solution is formed, which is hydrochloric acid. In aqueous solution, the hydrogen chloride molecule dissociates into hydrogen ions (H+) and chloride ions (Cl-):

The solution is called hydrochloric acid. Hydrochloric acid reacts with metals, metal oxides, hydroxides (soluble bases) and metal carbonates..
This suggests that when hydrogen chloride gas is dissolved in water an acidic solution is formed, for example:
- it turns damp blue litmus paper red;
- it reacts with various substances just like other acids (see table bellow); and
- it conducts electricity, yielding hydrogen gas at the cathode and chlorine gas at the anode.
Reactions of aqueous hydrogen chloride
| Acid reacting with | General equation |
| oxide (base) | acid + metal oxide ® salt + water |
| alkali (soluble base) | acid+metal hydroxide(akali)®salt+water |
| metal | acid + metal ® salt + hydrogen |
| metal carbonate | acid + metal carbonate®salt+water+CO2 |
The Uses of Hydrogen Chloride
Explain the uses of hydrogen chloride
These are:
- It is chiefly used in the production of hydrochloric acid. When the gas is dissolved in water in the appropriate proportions, hydrochloric acid is formed.
- Aqueous hydrogen chloride is used in qualitative and quantitative analysis.
- It is an important reagent in other industrial chemical transformations, e.g. hydrochlorination of rubber and production of vinyl and alkyl chlorides.
- In the electronics industry, it is used to both rub semiconductor crystals and to purify silicon.
- It is used in the textile industry, to separate cotton from wool and fluff.
- In the laboratory, anhydrous forms of the gas are particularly useful for generating chloride-based Lewis acids.
- It is used to remove rust from the oxidized metals.
- It is extensively used in the manufacture of medicines and is a key substance utilized to turn maize and other agricultural products into artificial sweeteners.
Sulphur
Occurrence
Sulphur is a yellow, crystalline, non-metallic solid. Its symbol is S. It has an atomic number of 16. Sulphur exists in nature as a free element and in compounds.
As a free element, sulphur is found in several countries such as Italy, Mexico, Japan, Poland, USA and Sicily. In its combined state, sulphur is found combined with metal ores such as galena (PbS), iron pyrites (FeS2), Copper pyrites (CuFeS2) and zinc blend (ZnS). It is also found in natural gas as hydrogen sulphide (H2S) and in crude oil as organic sulphur compounds.
The Extraction of Sulphur from Natural Deposits
Describe the extraction of sulphur from natural deposits
Sulphur is extracted from its underground deposits by the Frasch process. The Frasch process makes use of the low melting point (119oC) of sulphur.
In this process, a hole about 30 cm in diameter is bored down through the clay, sand, and limestone to the sulphur beds. This boring is lined with an iron pipe and inside the pipe, is sunk a device called sulphur pump. The pump consists of three concentric pipes (cylindrical pipes with a common centre) which end in a reservoir of a large diameter (see figure bellow).
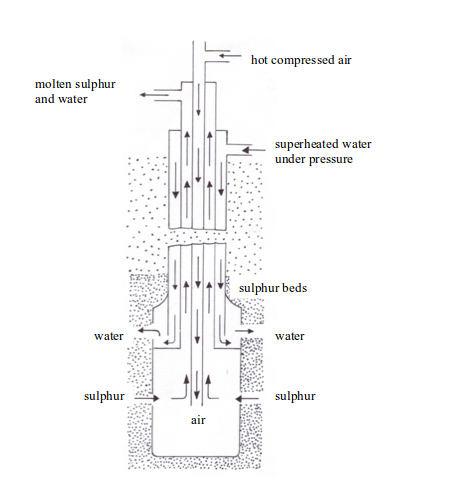
Extraction of sulphur
Down the outermost of the three pipes is forced a stream of water at about 170oC. This water must be kept at a pressure of about 10 atmospheres to maintain it in the liquid state, i.e. it is superheated water, and it is hot enough to melt the sulphur. The molten sulphur flows into the reservoir at the base of the pump and is forced up to the surface. The sulphur obtained is 99.5% pure and can be used without any purification.
The stages involved in the entire extraction process can be summarized as follows:
- Superheated water (170oC) is pumped through the outer pipe to melt the sulphur.
- Hot compressed air (10 atm) is pumped down through the inner pipe. The combination of the hot water and the hot air melts the sulphur. The molten sulphur, hot air and hot water form a froth.
- The froth is forced to the earth’s surface through the middle pipe by the compressed air.
- The molten sulphur is collected in large tanks (where the water drains off), cooled and solidified.
The Properties of Sulphur
Explain the properties of sulphur
Sulphur is a relatively reactive element that readily reacts with other elements to form compounds such as oxides, chlorides and sulphides.
In chemical reactions, sulphur exhibits both oxidizing and reducing properties.
1. Oxidizing properties of sulphur
Heated sulphur reacts with metals such as iron, copper, zinc and tin to give metal sulphides.
When a mixture of iron filings and powdered sulphur, in the proportion of 56 to 32, that is 7:4 (the ratio of their relative atomic masses), is heated, the two react in a highly exothermic reaction. The heat given out makes the mixture to continue glowing even after the heating has stopped. A black or dark grey iron (II) sulphide is formed.

In this reaction, sulphur acts as an oxidizing agent which oxidizes iron metal to iron (II) ion.

Sulphur is reduced to sulphur ion

Iron (II) sulphide is not attracted by a magnet since it is not magnetic. The magnetic property of iron is lost when this compound is formed.
The copper powder reacts with sulphur in the same way to form copper (II) sulphide.

2. Reducing properties of sulphur
Dilute acids do not act upon sulphur. However, sulphur is oxidized by hot concentrated sulphuric acid with the formation of sulphur dioxide.
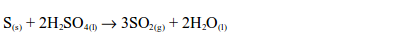
In this reaction, the sulphur is oxidized by the acid to sulphur dioxide and the acid is reduced to the same substance.
The filter paper soaked in potassium dichromate changes from orange to green. The sulphur dioxide reduces chromium from chromium (VI) to chromium (III).

When sulphur reacts with hot concentrated nitric acid, brown fumes of nitrogen dioxide are formed.
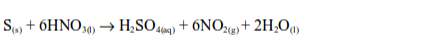
Sulphur is oxidized by hot concentrates nitric acid to sulphuric acid while nitric acid is reduced to nitrogen dioxide and water.
The Uses of Sulphur
Explain the uses of sulphur
Includes
- Most of the sulphur produced in the world (90%) is used to manufacture sulphuric acid. Sulphuric acid is an important reagent in many industrial processes.
- Sulphur is used in the manufacture of sulphur dioxide (used in the Contact Process for the manufacture of sulphuric acid).
- Manufacture of calcium hydrogensulphite, Ca(HSO3)2, and sodium sulphite which are used for bleaching wood straw and wood pulp in the paper industry.
- It is also used for vulcanization of natural rubber. Rubber is usually sticky and soft. When heated with sulphur (vulcanization), it becomes hard and strong.
- It is used for dusting vines to prevent growth of certain kinds of fungi and also as an insecticide.
- Sulphur is used in smaller quantities for the manufacture of dyes, explosives, fireworks, gunpowder etc. For example gunpowder is a mixture of potassium nitrate, carbon and sulphur.
- It is used in the manufacture of various organic compounds such as plastics and pharmaceuticals like sulphur ointments e.g. sulphadimadine, septrin e.tc.
- Photographic chemicals such as carbon disulphide (CS2) and sodium thiosulphate (Na2S2O3) are made using sulphur as one of the raw materials.
- Some is added to cement to make sulphur concrete. Unlike ordinary cement, this is not attacked by the acid. So it is used for walls and floors in plants where acid is used.
Sulphur Dioxide
Preparation of sulphur dioxide
In the laboratory, sulphur dioxide is prepared by heating a mixture of sodium sulphite and dilute hydrochloric acid. The reaction equation is:
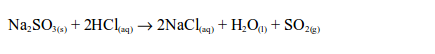
Alternatively, sulphur dioxide can be prepared by heating a mixture of concentrated sulphuric acid and copper. In this case, there is no reaction until the mixture in the flask becomes hot. Then rapid effervescence occurs and the gas is usually collected as shown.
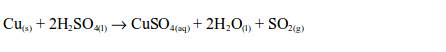
A dark brown mixture is left in the flask. It contains anhydrous copper (II) sulphate and certain impurities.
The Properties of Sulphur Dioxide
Describe the properties of sulphur dioxide
Physical properties
These are:
- The gas is colourless with an irritating (pungent), chocking smell.
- It is denser than air. Its density is 2½ times that of air.
- It is readily soluble in water and forms an acidic solution of sulphurous acid.
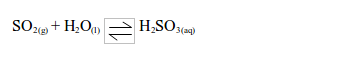
Acidic characteristics of sulphur dioxide
As indicated above, sulphur dioxide gas dissolves in water to form an acid solution of sulphurous acid, H2SO3. The solution turns blue litmus paper red. Sulphur dioxide is thus an acidic gas.
Chemical properties
The reaction characteristics described below explains the chemical properties of sulphur dioxide gas.
The reducing properties of sulphur dioxide
(i) Reduction of dyes in flower petals or colour in paper (bleaching)
Sulphur dioxide bleaches the colours in dyes such as flower pigments. When the flower pigments or dyes contain oxygen, they are coloured. Sulphur dioxide reduces the dye (removes oxygen from it) and the dye, therefore, turns colourless. This process can be summarized as follows:
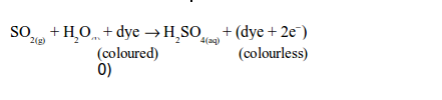
In this process, sulphur dioxide dissolves in water to form sulphurous acid:

Then the acid takes up oxygen from the dye of the flowers or paper and forms sulphuric acid. The removal of oxygen from the dye converts it to a colourless compound:
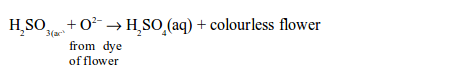
However, the oxygen from the air may oxidize the reduced colourless compound back to the original coloured compound.
(ii) Reduction of nitric acid
If sulphur dioxide gas is bubbled through concentrated nitric acid, brown fumes of nitrogen dioxide gas are formed:
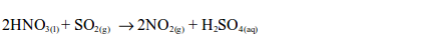
The nitric acid is reduced to nitrogen dioxide and the sulphur dioxide is oxidized to sulphuric acid.
(iii) Reduction of acidified potassium permanganate solution
Sulphur dioxide decolourized purple potassium permanganate solution.
Sulphur dioxide is first converted to a sulphite (SO32-), after reacting with water, and then oxidized to a sulphate (SO42-) by the permanganate.
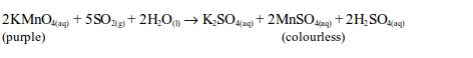
The ionic equation is:
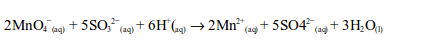
The oxidation state of manganese changes from +7 to +2. This is a reduction reaction.
(iv) Reduction of acidified potassium dichromate solution
When sulphur dioxide is mixed with potassium dichromate (VI) solution the orange colour of the solution changes to green. The dichromate (VI) is reduced to chromate (III) which is green in colour when in aqueous state:
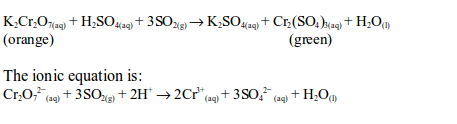
The oxidation state of chromium changes from +6 in potassium dichromate (VI) to +3 in chromic sulphate. On the other hand, sulphur dioxide is oxidized by the dichromate (VI) to sulphate (SO42-).
Other reduction reactions involving sulphur dioxide gas
Sulphur dioxide reduces chlorine, bromine and iodine to the hydrogen halides in the presence of water:
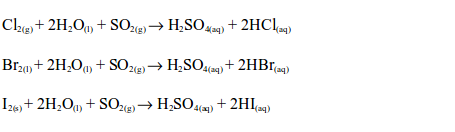
In all these cases, the solution changes from brown to colourless.
The oxidizing properties of sulphur dioxide
(i) Oxidation of hydrogen sulphide
Iron (II) sulphide reacts with dilute hydrochloric acid to produce hydrogen sulphide gas and iron (III) chloride:
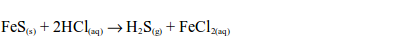
Hydrogen sulphide gas has a smell similar to that of a rotten egg. The hydrogen sulphide gas is oxidized by sulphur dioxide in the gas jar to sulphur:
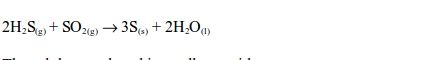
The sulphur produced is a yellow residue.The reaction takes place in the presence of moisture which acts as a catalyst. That is why water is added in the jar.
(ii) Oxidation of magnesium
When burning magnesium is lowered into a jar containing sulphur dioxide gas, a white solid, magnesium oxide, and yellow pieces of sulphur are formed:

Tests for sulphur dioxide gas
Includes
- The gas can be identified by its characteristic pungent and choking smell.
- It can also be detected by putting into it a filter paper that has been previously dipped into an acidified solution of potassium dichromate (VI). The colour of the filter paper changes from orange to green due to the reduction of dichromate (VI) to chromate (III).
- Sulphur dioxide also decolourized acidified potassium permanganate solution.
The Uses and Hazards of Sulphur Dioxide
Explain the uses and hazards of sulphur dioxide
Sulphur dioxide has got a number of uses in daily life. However, there are also some hazards which can be caused by the gas if its production is not controlled.
Uses of sulphur dioxide
The following are some uses of sulphur dioxide gas:
- The gas is used in the industrial manufacture of sulphuric acid in the Contact Process.
- It is used as a bleaching agent for wood pulp, silk, wool and straw.
- Its poisonous nature makes it a useful fumigant. So it is used in fumigation. The gas is poisonous to all organisms, particularly bacteria.
- It is used as a preservative and sterilizing agent in making soft drinks and jam, and in dried fruits. A very low concentration of the gas in food prevents fermentation as it stops the growth of bacteria and moulds. Its reaction with oxygen prevents oxidation of juices and other liquids to which it is added for preservation.
Hazards of sulphur dioxide gas
The hazards of sulphur dioxide gas are due to its effect in environmental pollution and the health problems accompanied with that pollution. Sulphur dioxide is a major air pollutant. The major sources of sulphur dioxide in the air are power plants that use fossil fuels such as coal, diesel and petrol; industrial boilers; and exhaust emissions from motor vehicles. The gas is also produced during metal smelting and other industrial processes.
Half of sulphur dioxide output comes from burning coal in coal-fired power stations. All coal contains small amounts of sulphur. So when the coal is burnt to produce energy, the sulphur in the coal reacts with oxygen in the air to produce sulphur dioxide

Sulphur dioxide is a very irritating gas and is thought to be the cause of bronchitis and other lung diseases. Exposure to higher concentrations of the gas can cause impairment of the respiratory function and heart diseases.
Sulphur dioxide also causes acid rain. This occurs when the gas comes in contact with moist air. The sulphur dioxide dissolves in water vapour from the clouds and combines with oxygen from the atmosphere to form an acid – sulphuric acid:
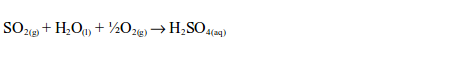
Acid rain damages the leaves and barks of plants making them more vulnerable to diseases, weather and insects. When acid rain reaches the lake, river or other water bodies it makes the whole water body acidic. Even a low concentration of acid in the water can kill fish and other marine organisms.
Sulphuric Acid
The Contact Process for the Manufacture of Sulphuric Acid
Describe the contact process for the manufacture of sulphuric acid
Sulphuric acid is an important laboratory and industrial reagent. It is produced in large scale through the Contact Process. The process involves four major stages. These are:
- production of sulphur dioxide;
- purification of sulphur dioxide;
- catalytic conversion of sulphur dioxide (SO2) to sulphur trioxide (SO3); and
- conversion of sulphur trioxide to sulphuric acid.
1. Production of sulphur dioxide
The sulphur dioxide used in the Contact Process can be obtained from different sources. These include:
(a) Burning sulphur in air:

This is the most convenient method of producing sulphur dioxide.
(b) Burning sulphide ores such as iron pyrite (FeS2) and zinc blend (ZnS):

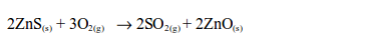
Sulphur dioxide gas is produced as a by-product.
2. Purification of sulphur dioxide
The sulphur dioxide produced in the first stage is mixed with air, ready for passing it over the catalyst. Before contact with the catalyst is allowed, the gas mixture has to be purified to remove impurities. This is achieved by passing the mixture through an electrostatic precipitator to remove any dust. It is then washed with water to remove impurities such as traces of arsenic (III) oxide (As2O3). The gas mixture is then passed through concentrated sulphuric acid to remove all moisture. The three impurities (As2O3, dust and moisture), if not removed, will poison the catalyst thereby rendering it useless.
3. Catalytic conversion of sulphur dioxide to sulphur trioxide
The purified and dried mixture of sulphur dioxide and air is passed through a heat exchanger to acquire the necessary heat for the conversion to sulphur trioxide. The mixture is then taken to the conversion chamber, which contains a catalyst. The catalyst used is finely divided vanadium (V) oxide (vanadium pentoxide, V2O5) which is heated to 450°C.
Originally, platinized asbestos was used as a catalyst. But, compared to vanadium (V) oxide, platinum is very expensive and easily poisoned by impurities. So it has been replaced by vanadium (V) oxide as the usual catalyst used in the Contact Process.
Sulphur dioxide remains in contact with the catalyst during the conversion process, hence the name Contact Process. The reaction that takes place during the conversion is:
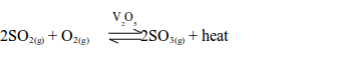
The conversion takes place at a temperature of 450°C and normal atmospheric pressure (1 atm).
The reaction is exothermic, which means that, as sulphur trioxide is formed, heat energy is released. If the temperature rises above 450°C the yield of sulphur trioxide decreases.
Once the reaction has started, no external heating is required. Thus, the heat exchanger maintains the temperature at 450°C. According to Le Chatelier’s principle, a lower temperature should be used to shift the equilibrium to the right, hence increasing the percentage yield. However, too low temperature will lower the formation rate to an economical level. Hence, to increase the rate, high temperature (450°C), medium pressure (1-2 atm) and a catalyst (V2O5) are used to ensure maximum yield. The catalyst only serves to increase the rate of reaction as it does not change the position of the dynamic equilibrium.
About 98% of the possible yield of sulphur trioxide is obtained.
4. Conversion of sulphur trioxide to sulphuric acid
The sulphur trioxide from the conversion chamber is passed through a heat exchanger to remove excess heat. It is then taken to an absorption tower where it is dissolved in concentrated sulphuric acid to form oleum or fuming sulphuric acid:

Oleum is then carefully diluted with the correct amount of water to give ordinary concentrated sulphuric acid:

Sulphur dioxide cannot be dissolved directly in water to form sulphuric acid. The reaction is so highly exothermic that the heat produced vapourizes the sulphuric acid formed. This makes it difficult to collect the gas because the acid vapour (mist) produced is very stable and cannot be condensed.
The flow diagram for the manufacture of sulphuric acid is show in the figure below
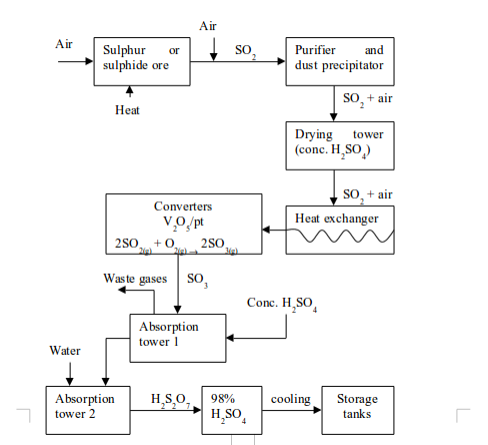
A flow diagram of the Contact Process
The Properties of Sulphuric Acid
Explain the properties of sulphuric acid
Chemical properties of dilute sulphuric acid
Dilute sulphuric acid reacts with metals, metal oxides, metal hydroxides and metal carbonates and hydrogencarbonates to produce salts.
Reaction with metals
Dilute sulphuric acid reacts with many metals above hydrogen in the activity series to form sulphates and hydrogen gas, e.g.
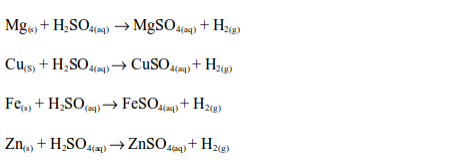
Reaction with metal oxides
Reactions of metal oxides with dilute sulphuric acid are neutralization reactions. Metal oxides react with dilute sulphuric acid to form a salt (sulphate) and water, e.g.
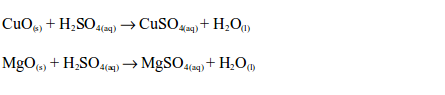
Reaction with metals hydroxides
The reaction between dilute sulphuric and a metal hydroxide is a neutralization reaction. Metal hydroxides react with dilute sulphuric acid to form a sulphate and water, e.g

Reaction with metal carbonates and hydrogencarbonates
Dilute sulphuric acid reacts with metal carbonates and hydrogencarbonates to give metal sulphates, carbon dioxide and water, e.g.

Chemical properties of concentrated sulphuric acid
Concentrated sulphuric acid as a dehydrating agent
As a dehydrating agent, it will remove the elements of water (hydrogen and oxygen) from a compound to form a new compound. It will dehydrate sugar, paper and wood. These are all made of carbon, hydrogen and oxygen. The acid removes the hydrogen and oxygen as water, leaving carbon behind.
When concentrated sulphuric acid is added to sugar, a vigorous reaction occurs, causing the reaction mixture to rise and fill the beaker.
The colour of the sugar changes to brown and finally black. Concentrated sulphuric acid dehydrates sugar (glucose) by taking away the elements of water (hydrogen and oxygen) from the sugar, leaving carbon.

The acid will also dehydrate sucrose to form carbon.

The final product is a black mass of carbon. The reaction is highly exothermic. The heat produced evaporates the water formed from the reaction.
The acid also dehydrates some hydrated salts. When concentrated sulphuric acid is added to hydrated blue copper (II) sulphate crystals, the colour changes from blue to white. The acid dehydrates the hydrated copper (II) sulphate crystals to anhydrous copper (II) sulphate powder:

Concentrated sulphuric acid also dehydrates methanoic acid and ethanol to give carbon monoxide and ethene gases respectively:
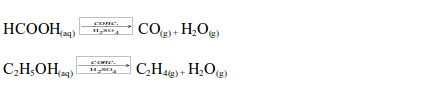
Concentrated sulphuric acid as a drying agent
As a drying agent, concentrated sulphuric acid absorbs traces of water from substances. Because of its ability to absorb water, it is used for drying most gases prepared in the laboratory that it would not react with. It cannot be used for drying ammonia, carbon dioxide, hydrogen sulphide or any gas with which it reacts.
Sulphuric acid as an oxidizing agent
Hot concentrated sulphuric acid is a strong oxidizing agent. It oxidizes both metals and non-metals while it is reduced to sulphur dioxide.
Concentrated sulphuric acid oxidizes charcoal (carbon) to carbon dioxide, sulphur to sulphur dioxide and copper to copper (II) sulphate.
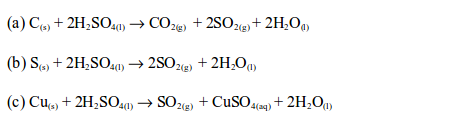
With sulphur and copper, the orange colour of the dichromate (VI) paper changes to green. This confirms the presence of sulphur dioxide gas.
With carbon, a white precipitate is formed on the glass rod when the rod dipped in lime water (calcium hydroxide) is placed in the mouth of the test tube. This confirms the presence of carbon dioxide gas which reacts with the calcium hydroxide on the glass rod to produce a white precipitate of calcium carbonate.

The Uses of Sulphuric Acid
Explain the uses of sulphuric acid
Sulphuric acid is one of the most important industrial chemicals. It has widely varied uses and plays some part in the production of nearly all manufactured goods. The following are some of the uses of sulphuric acid:
- Manufacture of fertilizers The major use of sulphuric acid is the production of fertilizers such as ammonium sulphate and superphosphates (phosphate fertilizers).
- Manufacture of chemicals It is widely used in the manufacture of chemicals e.g. in making hydrochloric acid, nitric acid, phosphoric acid, sulphate salts, synthetic detergents, soap, paints and pigments, explosives, plastics and drugs.
- Refining of crude oil A large quantity of sulphuric acid is used in refining crude oil.
- Extraction and manufacturing of metals Sulphuric acid is used in the iron and steel-making industry to remove rust and scale from the surface of the rolled iron sheets. It is also used in processing metals e.g. in pickling (cleaning) iron and steel before plating them with tin or zinc to produce galvanized iron.
- Manufacture of alum Sulphuric acid is used in the manufacture of aluminium sulphate, which is used in water treatment plants to filter impurities and to improve the taste of water. Aluminium sulphate is made by reacting bauxite with sulphuric acid.
- Manufacture of natural and man-made fibres Sulphuric acid is used for making natural and synthetic (artificial) fibres. For example, the textile called rayon is made from cellulose fibres derived from wood. These fibres are dissolved in a solution of tetraamminecopper (II) sulphate to produce a thick liquid which is then injected into sulphuric acid to form rayon fibres.
- Other uses:Sulphuric acid is used as (i) an electrolyte in lead-acid batteries, which are used in cars, to allow the flow of electrons between the plates in the battery. The sulphuric acid used in this way is called battery acid; (ii) as a general dehydrating agent in its concentrated form in tanning leather; and (iii) in waste water treatment.
Nitrogen
Occurrence
Nitrogen makes about 78% of the air by volume. The element also occurs combined with other compounds in the form of sodium nitrate, Chile saltpetre, NaNO3 (as a mineral deposit in Chile), and in the soil as ammonium sulphate, sodium nitrate, potassium nitrate and calcium nitrate.
Combined nitrogen also occurs as a constituent of all living matter of plants and animals in the form of proteins, enzymes, alkaloids.
Preparation of a sample of Nitrogen in the Laboratory
Prepare a sample of nitrogen in the laboratory
Nitrogen can be prepared in the laboratory by either separating the gas from the air or by heating ammonium nitrite.
(i) Preparation of nitrogen by the action of heat on ammonium nitrite
The reaction between sodium nitrite and ammonium chloride gives sodium chloride and ammonium nitrite.

A solution of ammonium nitrite readily decomposes, on slight warming, to give nitrogen gas.
As the solution becomes warm, rapid effervescence occurs as more nitrogen is given off.

The nitrogen evolved may be collected over water because the gas is only slightly soluble in water, at ordinary temperature, and slightly denser than air.
Tests for nitrogen gas
Aim: To test for the presence of nitrogen.
Procedure
Using the four gas jars of nitrogen gas collected in the previous experiment (Experiment 1.16), carry out the following tests for nitrogen gas and write down your observations:
- Remove the cover from the first jar and smell the gas. Observe the colour of the gas and identify its smell.
- Remove the cover from the second jar and put in it a piece of damp universal indicator.
- Place a lighted splint into the third gas jar.
- To the fourth jar, add some calcium hydroxide solution (lime water) and shake.
Observations and inferences
- The gas is colourless and odourless. This distinguishes it from gases such as sulphur dioxide, ammonia, hydrogen chloride, etc.
- The colour of the indicator does not change. This shows that nitrogen is a neutral gas.
- The lighted splint is extinguished and the gas does not burn. It can not, therefore, be any gas which supports combustion, e.g. oxygen, dinitrogen oxide, or any combustible gas, e.g. hydrogen sulphide, carbon monoxide, hydrogen, etc.
- After the above tests, the only gas with which nitrogen may be confused is carbon dioxide. To distinguish it from carbon dioxide, the gas is dissolved in calcium hydroxide solution. Nitrogen leaves the calcium hydroxide unchanged while carbon dioxide turns the solution milky (due to formation of a precipitate of CaCO3).
Properties of nitrogen
Physical properties
Nitrogen is colourless and odourless. It is slightly less dense than air and sparingly insoluble in water. The gas is neutral to litmus.
Chemical properties
Under ordinary conditions, nitrogen gas is inert. However, the gas only takes part in reactions at very high temperature as follows:
1. It reacts with red hot metals, like magnesium and calcium to form nitrites

2. Nitrogen does not burn nor does it support combustion. When heated, the gas combines with oxygen to form nitrogen monoxide gas:

3. At ordinary pressure and moderately high temperatures, nitrogen reacts with hydrogen in the Haber Process to produce ammonia:

The Uses of Nitrogen
Explain the uses of nitrogen
Here are some uses of Nitrogen:
- Manufacture of fertilizers: Nitrogen is used to manufacture nitrogenous fertilizers. These include diammonium phosphate (DAP), calcium ammonium nitrate (CAN), ammonium superphosphate (ASP), ammonium nitrate (AN), ammonium phosphate sulphate (APS), ammonium sulphate nitrate (ASN), ammonium sulphate (AS) and urea.
- Refrigeration: Because of its low boiling point (-196oC), liquid nitrogen is used as a refrigerant for storing organs in research laboratories, bull semen for artificial insemination, eggs and other cells for medical research and reproductive technology, etc. It is also used for preservation of food products and for their transportation.
- Processing reactive substances: Some reactions require an inert atmosphere in order to proceed as desired. Because of its low reactivity, nitrogen is used to provide an inert atmosphere for storing and processing reactive substances.
- Manufacture of nylon: Nitrogen is used in the manufacture of synthetic fibres such as polyamides. Polyamides are commonly known as nylons. Nylons are chemically inert and are stronger than natural fibres. They are used in making fishing nets, clothes, ropes, and many other items.
- Manufacture of ammonia: Nitrogen is used in the manufacture of ammonia through the Haber Process.In the Haber Process, ammonia is manufactured by direct combination of nitrogen and hydrogen.Nitrogen and hydrogen are mixed in the ratio of 1:3. The gases are then reacted together at a temperature of about 450°C and a pressure of 250 atmospheres in the presence of finely divided iron as a catalyst. N2(g) + 3H2(g) ⇔2NH3(g) + heat The gases are cooled while still under pressure and ammonia is removed as a liquid.
- Manufacture of nitric acid: The ammonia gas manufactured using nitrogen in the Haber Process is used in the manufacture of nitric acid by catalytic oxidation.
- Plant nutrition: When atmospheric nitrogen is fixed into the soil by bacterial actions, it becomes a nutrient to plants. Nitrogen fixation refers to the conversion of atmospheric nitrogen, by certain species of bacteria, into nitrites.
Ammonia
Preparation of a Dry Sample of Ammonia Gas in the Laboratory
Prepare a dry sample of ammonia gas in the laboratory
Ammonia is a binary compound of nitrogen and hydrogen. Ammonia gas is colourless and has a strong pungent and choking smell. It does not occur free in air, but exists in nitrogenous organic materials such as hoofs and horns of animals. The gas can be released by heating or burning these materials.
Laboratory preparation of ammonia
Ammonia is prepared in the laboratory by heating any ammonium salt with an alkali. In most cases, ammonium chloride and calcium hydroxide (the cheaper alkali) are used. Both are solid so they must be thoroughly ground first to give a very fine mixture so that the reaction can occur efficiently.
Ca(OH)2(s) + 2NH4Cl(s)→CaCl2(s) + 2H2O(l) + 2NH3(g)
Calcium hydroxide reacts with ammonium chloride to produce ammonia gas, calcium chloride and water. The flask is tilted to prevent any condensed water formed from running back into the hot flask, which might break it.
Ammonia gas is dried by passing it over quicklime because it reacts with all the usual drying agents. Concentrated sulphuric acid is acidic and would absorb the gas forming a salt e.g. 2NH3(g) + H2SO4(l) → (NH4)2SO4(s)
It reacts with calcium chloride, forming solid complex compounds, e.g. 8NH3(g) + CaCl2(s) → CaCl2.8NH3(s)
Ammonia is less dense than air and very soluble in water, so it is collected as shown by upward delivery (or downward displacement of air).
Ammonia is an alkaline gas and it turns wet, red litmus paper blue.
The Properties of Ammonia
Describe the properties of ammonia
Physical Properties of Ammonia
- It is a colourless poisonous gas with a strong chocking smell.
- It is less dense than air.
- It is easily liquefied by cooling to -33°C or by compressing it.
- It turns wet, red litmus paper blue as it is the only common alkaline gas
- It is very soluble in water forming alkaline solution. Ammonia has the highest solubility of all known gases. About 800 volumes of the gas dissolve in 1 volume of water at 15°C. The fountain experiment below demonstrates this solubility.
Chemical properties of ammonia
Reaction with air and oxygen
Ammonia will not normally burn in air but can be made to do so in two ways:
- Burning ammonia in an oxygen-rich atmosphere; and
- Use of a catalyst.
Burning ammonia in oxygen-rich air
When ammonia is burned in air enriched with oxygen, the products formed are nitrogen and water. 4NH3(g) + 3O2(g) → 2N2(g) + 6H2O(l)
Using a catalyst
The source of ammonia gas in this experiment is concentrated ammonia solution which gives off fumes of ammonia gas. If some concentrated ammonia solution is left in a stoppered flask for a few minutes, the flask will quickly become full of ammonia gas by diffusion.
The catalyst used is the metal platinum. The platinum coil is heated in a bunsen flame until it is red hot. It is then lowered into a flask containing ammonia. The coil continues to glow even though it is not being heated. This indicates that a chemical reaction is taking place. Near the top of the flask brown fumes can be seen.
The products of this reaction are nitrogen monoxide (nitrogen (II) oxide), which is a colourless gas, and water.
4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g)
However, as the nitrogen monoxide gas moves towards the neck of the flask and comes into contact with the air, it reacts with the oxygen in the air to form nitrogen dioxide gas which is brown in colour. Hence, the brown fumes are seen at the neck of the flask.
2NO(g) + O2(g) → 2NO2(g)
Reaction with copper
When ammonia gas is passed over heated copper (II) oxide, the gas is oxidized to nitrogen and water by the hot copper (II) oxide while the oxide is reduced to copper.
3CuO(s) + 2NH3(g) → 3Cu(s) + 3H2O(l) + N2(g)
Copper (II) oxide is black. During the reaction it is oxidized to copper which is reddish brown in colour.
Reaction with hydrogen chloride gas
When ammonia and hydrogen chloride are mixed, dense white fumes of ammonium chloride are formed.
NH3(g) + HCl(g) → NH4Cl(s)
This test is simply performed by dipping a glass rod into concentrated hydrochloric acid then holding the glass rod at the mouth of a gas jar containing ammonia gas.
The Uses of Ammonia
Explain the Uses of Ammonia
Manufacture of fertilizer
Since ammonia solution is alkaline, it reacts with acids to form ammonium salts which are used as fertilizers.
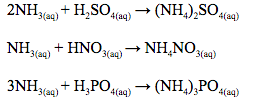
Other nitrogenous fertilizers manufactured using ammonia include ammonium sulphate nitrate (ASN), diammonium phosphate (DAP) and calcium ammonium nitrate (CAN)
Ammonia itself can be used directly as a fertilizer but has three disadvantages:
- Ammonia comes as a gas or a concentrated solution which is less easy to store; it is easier to store the solid ammonium salts.
- Ammonia is alkaline and can affect the natural pH of the soil.
- Ammonia easily evaporates if directly applied to the soil.
Manufacture of nitric acid
A lot of the ammonia from the Haber Process is used to make nitric acid. The raw materials for manufacture of nitric acid are ammonia, air and water.

The overall result is that ammonia is oxidized to nitric acid.
Cleaning
Ammonia solution is very useful in cleaning. This is because it softens water in homes and laundries and neutralizes acid stains caused by perspiration, hence making washing easier. It is also used as a grease solvent.
Refrigeration
Liquid ammonia is used in large-scale refrigerating plants such as in ships and warehouses. It used to be used in domestic refrigerators, but has now been replaced by non-toxic, non-corrosive chlorofluorohydrocarbons (CFHs).
Manufacture of synthetic fibres
In the textile industry, ammonia is used in the manufacture of synthetic fibres such as nylon and rayon.
Carbon
The Forms in which Carbon Exists
Name the forms in which carbon exists
Although carbon forms less than one percent of the earth’s crust by weight, it is the most interesting of all elements. This is because of the following reasons:
- All living things are made of carbon and its compounds.
- Over ¾ of the world’s power is obtained from carbon and its compounds.
- Over ¾ of all substances in the world are made of carbon.
Carbon is not commonly found in a free state. The free element is mainly found as graphite and diamond. Carbon occurs in a number of other forms, e.g. wood charcoal, animal charcoal, coke, soot (lampblack).
Carbon compounds are found in many naturally occurring substances such as coal, petroleum, wood, coal gas, natural gas, carbonates, shells, organic matter of all kinds, all living things, and occurs in the air to a small, but very important extent (0.03 – 0.04% by volume) as carbon dioxide. The carbohydrates, proteins and lipids in all living things contain carbon.
Pure carbon is found in the form of diamond in Tanzania (Mwadui), Sierra Leone, India, South Africa, Russia and South America; and impure carbon, as graphite, is found in Sri Lanka.
Allotropic Forms of Carbon
Describe allotropic forms of carbon
An element that exists in more than one form is said to exhibit allotropy. Allotropy is the existence of an element in more than one form (without change of state). The various forms are known as allotropes.
Allotropes of a given element differ in their physical properties and may differ in some chemical properties as well. The allotropic forms of carbon are:
- graphite;
- diamond; and
- amorphous carbon.
These allotropes have got different molecular structures. The structural differences are mainly due to the way their atoms are packed.
Graphite
Graphite has a layer structure. Figure 1.18 illustrates one layer of the structure of graphite. Each layer consists of carbon atoms covalently bonded together into hexagonal rings. These rings form flat parallel layers, one over the other. The force that hold the carbon atoms together are very strong. Adjacent layers are held together by weak van der Waals’ forces as shown in figure 1.19. The layers readily slide over one another, accounting for the soft and greasy texture of graphite.
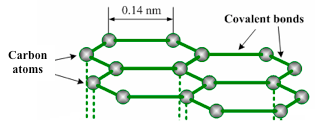
Carbon atoms in one graphite layer
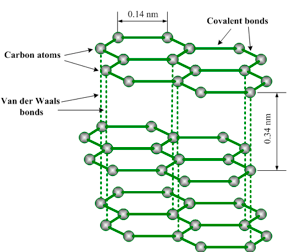
Graphite structure
Carbon atom has got four electrons in its outer shell. Each carbon atom forms three covalent bonds to other carbon atoms. Thus, three of its four outermost electrons are paired up to form covalent bonds. The fourth electron is not attached to any particular atom (delocalized) and is free to move anywhere along the layers. Graphite conducts electricity in the plane of the layers but not at right angles to them.
Because conduction of electricity involves movement of unshared electrons from one atom to another atom, graphite is a good conductor of electricity since the hexagonal layers permit this movement. It is also a good conductor of heat for a similar reason.
Physical properties of graphite
- It is a black, soft and slippery substance. It feels soapy and greasy. It has a metallic lustre and is opaque to light.
- It has low relative density (2.3) as compared to diamond (3.5)
- Graphite is a good conductor of heat and electricity due to the delocalized electrons.
- It has a very high melting point (3730°C) and boiling point (4830°C). The melting and boiling points are high because of strong covalent bonds between the carbon atoms which require more energy (heat) to break in order to melt graphite.
Uses of graphite
- It is used as an electrode in electrolysis and as a positive terminal in dry cells. The use of graphite as electrode in electrolysis has an advantage because it does not react readily with most substances (it is an inert electrode).
- It is used as a lubricant, particularly when high temperatures are involved, where the usual lubricating oils easily decompose due to extreme heat. It is a lubricant for dynamos, electric motors and fast-moving parts of machinery.
- The major use of graphite is in making lead pencils of different hardness, by mixing it with different proportions of clay. The weakly held layers of carbon atoms in graphite easily slide over each other and are left behind on paper as black marks.
- Being resistant to chemicals and having a high melting point and also because it is a good conductor of heat, graphite is used to make crucibles.
- Graphite has the ability to absorb fast-moving neutrons, thus, it is used in nuclear reactors to control the speed of the nuclear fission reaction.
Diamond
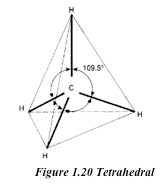
The basic unit of the diamond structure is shown in figure 1.20. Each carbon atom is covalently bonded to four other carbon atoms. This basic unit is repeated in three dimensions as shown in figure 1.21 to form a giant tetrahedral structure of millions of carbon atoms, all forming four covalent bonds to each other. The melting point of diamond is high. This is because of the strong covalent bonds between carbon atoms, which require a large amount of heat energy to break up.
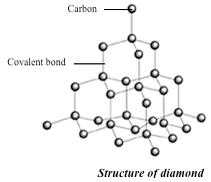
Physical properties of diamond
- It is the hardest natural substance known. This due to the strong covalent bonds between the carbon atoms in diamond. Again the compact tetrahedral arrangement of carbon atoms contributes to its hardness.
- It has the highest melting point (3550°C) and boiling point (4289°C).
- It has a high relative density (3.5) compared to graphite (2.3)
- It is a poor conductor of heat and electricity. This is because there are no free electrons to conduct electricity. All electrons are firmly held in covalent bonds.
- It is colourless, transparent and has a dazzling (amazing) brilliant lustre and radiance.
- It has a high refractive index of 2.5. The high refractive index results in high dispersion of light, making it suitable for use in jewellery.
Uses of diamond
- It is used in making jewellery.
- Due to its extreme hardness, it is used to make glass cutters, drilling devices, rock borers, and as an abrasive for smoothing very hard materials.
Comparison of the properties of diamond and graphite
| Diamond | Graphite |
| 1. Colourless, transparent andglittering | Black, opaque with metallic lustre |
| 2. Hardest natural substanceknown, used to cut glass andin drills | Soft to touch, greasy or soapy, can be used as a lubricant and in making lead pencils |
| 3. High relative density (3.5) | Low relative density (2.3) |
| 4. Non-conductor | Good conductor of heat and electricity |
| 5. Burns in air least readily (atabout 900°C) | Burns in air readily (at 700°C) |
| 6. Have strong C-C covalentbonds arranged octahedrally toform a giant molecular crystal | Have strong C-C bonds within the hexagonal rings in the sheets but weak Van der Waal’s forces in between layers. |
| 7. Its cleavage is difficult and itoccurs along the boundaries ofthe octahedral crystal unit | Cleavage easy, and occurs along the sheets or the layers. |
| 8. Prepared from graphite at very high pressure and temperature | Prepared from coke and silica mixture at high temperature |
| 9. Not attacked by potassiumchlorate and nitric acid together | Attacked by these reagents |
A proof that diamond and graphite are both allotropic forms of carbon can be shown experimentally by burning equal masses of each allotrope in a stream of oxygen. It is found that equal masses of each allotrope produce equal masses of carbon dioxide. C(s) + O2(g) → CO2(g)
The weight of carbon dioxide produced can be determined by allowing it to be absorbed in a weighed amount of potassium hydroxide solution. 2KOH(aq) + CO2(g) → K2CO3(aq) + H2O(l). Then, the weight of carbon dioxide is obtained by calculations based on the equation above.
Amorphous carbon
Amorphous carbon is carbon that does not have any clear shape, form or crystalline structure. Amorphous carbon is made of tiny bits of graphite with varying amounts of other elements considered as impurities. It is formed when a materials containing carbon is burned in a limited supply of oxygen, resulting in incomplete combustion.
Amorphous carbon exists in many forms. The following are the major forms in which it occurs:
- Charcoal
- Lampblack (soot) or carbon black
- Coke
Charcoal
Charcoal is made by heating organic material (animal or plant parts) to a high temperature in the absence of air or in the presence of limited amounts of oxygen or other reagents, catalysts, or solvents. This process is called destructive distillation. There are three categories of charcoal, namely, wood charcoal, animal charcoal and sugar charcoal.
Wood charcoal
Wood charcoal is made by heating wood or other vegetable matter (for example coconut shells) in the almost complete absence of air. Wood charcoal is light, porous and is a remarkably good absorbent for liquid or gases (1 cm3 of wood charcoal will absorb 100 cm3 of ammonia gas at 0°C).
Uses of wood charcoal
- Because of its ability to absorb large amounts of gas or liquid, it is used in gas masks to absorb poisonous gases in air in industrial process to recover volatile materials from waste gases.
- Wood charcoal can be used in metal refining instead of coke.
- Wood charcoal is a good source of energy. Thus, it is used as fuel for cooking and heating in homes.
Animal charcoal
Animal charcoal is made by heating animal bones in the absence of air. Its main component is calcium phosphate, Ca3(PO4)2, and carbon constitutes about 10% of the components.
Uses of animal charcoal: It is used in sugar industries to remove the colouring matter from brown sugar to make it white
Sugar charcoal
Sugar charcoal is a very pure form of carbon, and is made by removing the elements of water (oxygen and hydrogen) from sugar. This is achieved by using strong dehydrating agents such as concentrated sulphuric acid or concentrated nitric acid, which removes water from sugar.
Uses of sugar charcoal: Sugar charcoal is chiefly used in the manufacture of artificial diamonds.
Lampblack/soot/carbon black
This is produced by burning petroleum or any hydrocarbon in a limited supply of air (or in chlorine). It can also be produced from the burning of organic material at home. It is commonly found in the kitchen chimneys, lamps and on bases of cooking pans and pots.
Uses of lampblack/soot/carbon
- It is used in making printers’ ink, shoe polish and carbon papers.
- It is an important industrial material in the manufacture of tyres. It is used as a filler material in tyres.
Coke
Coke is made by destructive distillation of coal. During the process, coal is obtained as the major product. The other products formed when coal is destructively distilled are coal gas, coal tar, coal oil, gas carbon and ammonia liquor.
Uses of coke
- Coke is used as a fuel and as a reducing agent in the extraction of iron, lead and zinc. It is also used as a fuel in boilers.
- It is used in the manufacture of producer gas and water gas.
Chemical properties of carbon
- Carbon burns in excess oxygen to form carbon dioxide C(s) + O2(g) → CO2(g).In insufficient oxygen, carbon monoxide is formed.2C(s) + O2(g) → 2CO(g)
- Carbon has got reducing properties and thus acts as a reducing agent. Carbon reduces oxides of metals below it in the electrochemical and activity series to their respective metals. This occurs on strong heating, and this reaction is used industrially for extraction of metals from their ores:PbO(s) + C(s) → Pb(s) + CO(g);Fe2O3(s) + 3C(s) → 2Fe(s) + 3CO(g);ZnO(s) + C(s) → Zn(s) + CO(g).
- Sulphur vapours react with red hot carbon to give carbon disulphide. C(s) + 2S(g) → CS2(l)
- Carbon dioxide is reduced by red hot carbon to carbon monoxide C(s) + CO2(g) → 2CO(g);This reaction is used in the industrial manufacture of producer gas.
Carbon Dioxide
Preparation of a Dry Sample of Carbon Dioxide Gas in the Laboratory
Prepare a dry sample of carbon dioxide gas in the laboratory
Carbon dioxide is one of the oxides of carbon. The gas is present in the air at a level of approximately 0.03% by volume. It is also found dissolved in water. The gas is one of the by-products of all decaying organic matter. Without carbon dioxide there is no life on earth. It is used by all plants in the process of photosynthesis and both plants and animals evolve carbon dioxide in respiration.
Laboratory preparation of carbon dioxide
Carbon dioxide is prepared in the laboratory by the action of dilute hydrochloride acid on marble (calcium carbonate).
When dilute hydrochloric acid is poured on marble chips, effervescence occurs. Dilute hydrochloric acid reacts with the marble chips to give calcium chloride, water and carbon dioxide.
CaCO3(s) + 2HCl(aq) → CaCl2(g) + H2O(l) + CO2(g)
The Properties of Carbon Dioxide
Analyse the properties of carbon dioxide
Physical properties
- Carbon dioxide is a colourless and odourless gas.
- It is denser than air.
- When the gas is cooled to –78°C, it turns straight into the solid (it sublimes). Sublimation is the change of a gas straight into a solid or change of a solid straight into a gas. Solid carbon dioxide is called dry ice. It sublimes when it is heated or exposed to air.
- It has a melting point of –199°C and boiling point of –91.5°C.
- Carbon dioxide does not support combustion. This is why it is used in fire extinguishers.
Chemical properties
Reaction of carbon dioxide with lime water (Test for carbon dioxide)
When a little carbon dioxide gas is bubbled into lime water (calcium hydroxide solution), the solution turns milky. This is due to the formation of a white precipitate of insoluble calcium carbonate.
Ca(OH)2(aq) + CO2(g) → CaCO3(s) + H2O(l)
This is a confirmatory test for the presence of carbon dioxide. The test serves to distinguish carbon dioxide from any other gas.
When excess carbon dioxide is bubbled into the lime water, the white perceptible dissolves to form a clear solution of soluble calcium hydrogen carbonate: CaCO3(s) + H2O(l)+ CO2(g) → Ca(HCO3)2(aq)
Barium hydroxide can also be used to test for carbon dioxide as it forms a precipitate of barium carbonate: Ba(OH)2(aq) + CO2(g) → BaCO3(s) + H2O(l)
Reaction of carbon dioxide with magnesium
When a burning magnesium ribbon is lowered into a gas jar containing carbon dioxide gas, it continues to burn for a short time with a spluttering flame. A white ash of magnesium oxide and black specks of carbon are formed. The black specks of carbon can be seen on the sides of the gas jar.
2Mg(s) + CO2(g) → 2MgO(s) + C(s)
This clearly shows that carbon dioxide contains carbon and oxygen.
Reaction of carbon dioxide with water
Carbon dioxide reacts with water to form a weak carbonic acid. When carbon dioxide is bubbled into water, it dissolves to form a weakly acidic solution of carbonic acid:
H2O(l) + CO2(g) ⇔ H2CO3(aq)
The solution turns a blue litmus paper pink. This indicates that the solution is slightly acidic and hence too weak to turn the blue litmus paper to red (as strong acids do). The solution has no effect on red litmus paper.
The Uses of Carbon Dioxide
Explain the uses of carbon dioxide
Uses of Carbon Dioxide include:
- Fire extinguisher: Carbon dioxide is inert (i.e. it does not burn). It is dense than air and does not support combustion. Hence, it is a very useful fire-fighting chemical. When applied to fire, it forms a blanket over the burning material. Thus, it prevents air (oxygen) from reaching the burning material and therefore, extinguishing the flames.
- Manufacture of aerated (fizzy) drinks: Soda water and mineral water contain carbon dioxide dissolved under pressure. Because the gas is only slightly soluble, it is bubbled into these drinks under pressure to make more of it dissolve. When the bottles are opened, the gas escapes and it causes the “fizzy”.Dissolved carbon dioxide is responsible for the pleasant taste of soft drinks such as lemonade, Coca cola, Pepsi cola and other aerated drinks and mineral waters. Other beverages like beers are also bottled together with carbon dioxide.
- Refrigeration: Carbon dioxide is used for refrigeration purposes (i.e. in the deep-freezing of foods). The gas liquefies at ordinary pressure to form dry ice which sublimes at -78°C. Dry ice is a good refrigerant because it leaves no liquid after sublimation as is the case with ordinary ice.
- Manufacture of sodium carbonate by the Solvary Process:Carbon dioxide is used in the manufacture of anhydrous sodium carbonate in the Solvary Process. The sodium carbonate produced is used in the manufacture of glass.
- Manufacture of baking soda: Carbon dioxide is used in making baking soda (sodium bicarbonate). Baking soda is prepared by passing carbon dioxide into cold concentrated sodium hydroxide solution: CO2(g) + 2NaOH(aq) → Na2CO3(aq) + H2O(l).Further addition of carbon dioxide produces sodium bicarbonate which, at sufficiently high concentration, will precipitate out of the solution as a solid: Na2CO3(aq) + CO2(g) + H2O(l) → 2NaHCO3(s) Yeast and sodium bicarbonate (hydrogencarbonate) are important in the baking industry. Thus in baking of bread, yeast is added to flour, sugar and water (forming the dough). In the making of cakes, baking powder (a mixture of bicarbonate and an acid) is used instead of yeast.
- Rain making: When pieces of dry ice (solid carbon dioxide) are dropped into clouds, the temperature of the clouds is lowered to such an extent that rain precipitates out.
- Photosynthesis: Plants use carbon dioxide from the air to manufacture their own food through the process of photosynthesis.







EmoticonEmoticon